Soil Acidity
Introduction
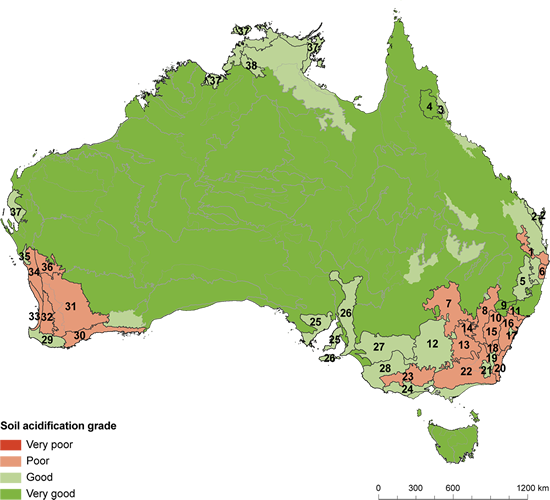
FIgure 1: A map of Australia produced by the Department of the Environment (Australia) in 2011 in a report concerning the problem of soil acidification in Australia. As this map shows it is very poor in areas with heavy farming such as New South Wales and Western Australia. Source: http://www.environment.gov.au/science/soe/2011-report/5-land/2-state-and-trends/2-2-soil
Nitrogen is very important to the plants growth however since the establishment of farms and the use of fertiliser, excess nitrogen has been released into our soils. This has been causing soil acidification. Soil acidity is a major environmental and economical problem that is considered to farmers as one of the major problems they have to face. When the pH (a measure of the concentration of hydrogen atoms) is under 5.5 in the topsoil or 4.8 in the subsurface, it is considered to be experiencing soil acidification. Acidification normally starts occuring on the upper layers of the soil and works its way down, getting more expensive to treat and reducing crop productivity the further down the problem occurs. It is estimated that approximately 50% of Australia's farmlands (50 million hectares) is affected by this problem. As figure 1 shows, the areas that have been surveyed, 70% has been classified as poor by the Australian Government. Acidic soil causes a significant reduction in productivity as crops grow slower because most crops cannot tolerate high acidic levels, some cannot even survive. There is also a wider impact as the polluted runoff may poison animals or humans utilizing the water source.
Causes
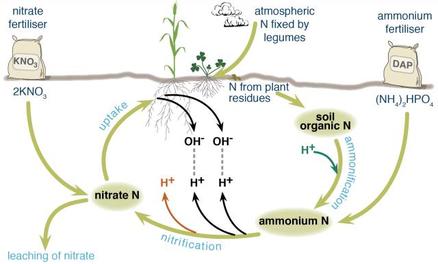
Soil acidity is a natural process as nitrogen is recycled through the death and birth of the plants however agriculture has accelerated this process making it unbalanced. It is mainly caused by nitrate leeching of the soil due to the application of ammonia-based fertilisers. As figure 1 shows, when ammonium fertiliser is applied and breaks down into negatively-charged nitrate, nitrification occurs by transforming the nitrate to nitrite by a chemical reaction (bonding) involving positively-charged hydroxide to be used by the plants however most often there is the lack of hydroxide (released from plants) for the nitrate to bond onto. This excess nitrate is leached into the soil resulting in the decrease of the pH. The soil in Western Australia is very prone to nitrate leeching as there is not enough positively-charged hydroxide in the soil to be bonded with the nitrate and this has meant that soil acidity is a very pressing problem in the state affecting most farmlands as figure 1 shows.
Another cause of soil acidity is the removal of plants. Most of plant material is slightly alkaline and contains nitrate ions. Once these plants are removed, the nitrate ions are released back into the soil and the slightly alkaline effect of the plant is removed hence the soil pH becomes slightly more acidic. This process is normally repeated many times when clearing out land, stacking the effects.
Effects
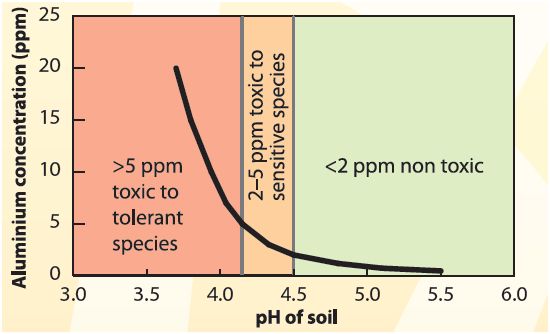
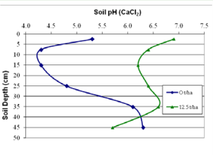
The main effect of soil acidification is aluminium toxicity. When the pH of the soil drops, the aluminium in the soil becomes soluble (able to be mixed with water) and in this form, the aluminum slows down root growth which in turn restricts water and nutrient access for the plant. This results in poor crop yield/production which is the result of the limited water and nutrient available to the plant and this effect is the most obvious during drying seasons when the water is in the subsoil requiring deep roots which aluminium intoxicated plants lack. Figure 3 demonstrates the relationship between the pH of the soil and the concentration of aluminium in the soil. Between the pH of 5.5 and 4.5 the aluminium is below 2.5 ppm however past this, the aluminium concentration increases exponentially and becomes too toxic for any plants to survive. As figure 3 and 4 shows, an aluminium concentration of 15 ppm or 4 pH drastically alters the plant's structure especially the root growth. The roots of the plant on the right is much shorter than the plant to the left in figure 4. Figure 5 is a close examination of the root structure of an aluminum poisoned plant as shown on the right photograph. The root is deformed and brittle, reducing the surface area exposed to the soil thus reducing the nutrient intake while the healthy root on the left is immaculate, retaining a strong structure and ensuring the maximum surface area is exposed to the soil with the shape of a cone.
Effective Solutions
 Figure 9: A photograph of a tractor pulling a spader along an area requiring delving. The spader mixes the clay in the subsoil with the topsoil. Source: http://www.agbureau.com.au/projects/soil_acidity
/ABA_WS_1aa_Final_-_Identification_and_treatment.pdf
Figure 9: A photograph of a tractor pulling a spader along an area requiring delving. The spader mixes the clay in the subsoil with the topsoil. Source: http://www.agbureau.com.au/projects/soil_acidity
/ABA_WS_1aa_Final_-_Identification_and_treatment.pdf
Delving: Delving the soil is essentially mixing the topsoil and the clay in the subsoil. It only works without additional lime when the pH of the subsoil is less acidic than the pH of the topsoil. It has been proven that the utilization of this method results in better plant growth. The pH of the subsoil should be tested before the utilization of this method as if the pH of the subsoil is lower than the topsoil and delving is commenced, it would be counterproductive.
If the pH of the topsoil is above 5.5 and the pH of the subsoil is below that of the topsoil, liming the topsoil then delving would allow the lime to mix with the subsoil as well. This would lead to faster recovery of the soil.
Both of these types of delving are done by a Spader. A spader is a large rotational hoe that mixes the soil up to 40 cm deep. Delving is the most effective measure however it requires expensive machinery and lost time that can be spent growing crops or grazing farm animals.
Using non-acidifying fertiliser: While fertilisers containing ammonia greatly increase the rate of acidification of the soil, fertilisers that contain calcium nitrate or potassium nitrate do not acidify the soil. These fertilisers even have a slight alkalizing effect. These fertilizers are now often used for horticulture crops or plants and prevents acidification.
If the pH of the topsoil is above 5.5 and the pH of the subsoil is below that of the topsoil, liming the topsoil then delving would allow the lime to mix with the subsoil as well. This would lead to faster recovery of the soil.
Both of these types of delving are done by a Spader. A spader is a large rotational hoe that mixes the soil up to 40 cm deep. Delving is the most effective measure however it requires expensive machinery and lost time that can be spent growing crops or grazing farm animals.
Using non-acidifying fertiliser: While fertilisers containing ammonia greatly increase the rate of acidification of the soil, fertilisers that contain calcium nitrate or potassium nitrate do not acidify the soil. These fertilisers even have a slight alkalizing effect. These fertilizers are now often used for horticulture crops or plants and prevents acidification.